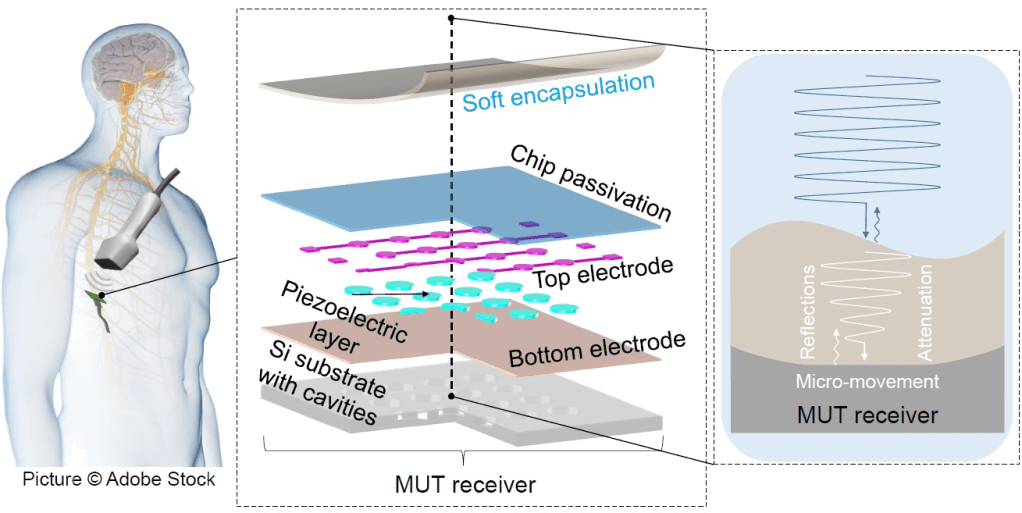
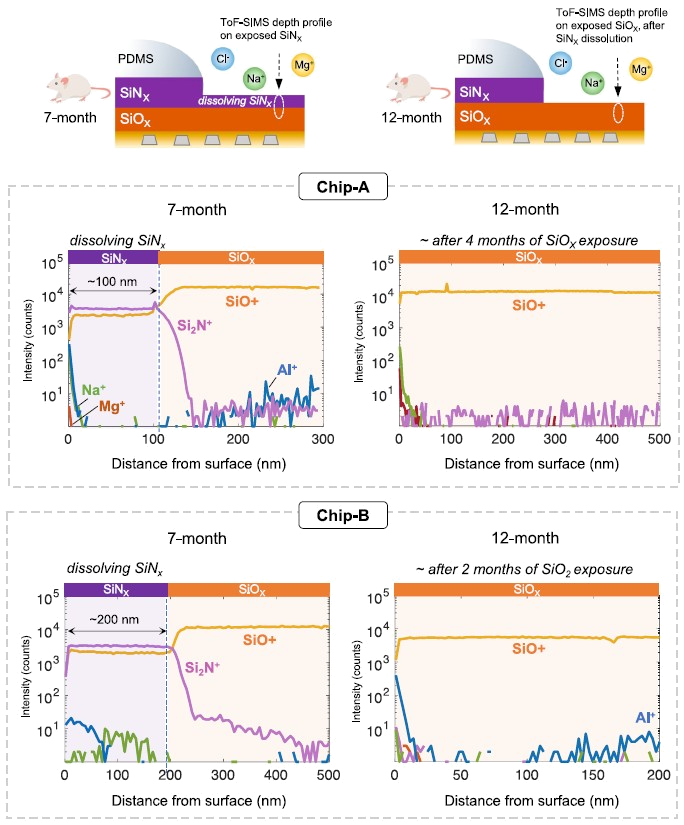
We are very excited to share our latest publication on the topic of Ultrasound transparent neural interfaces for multimodal interaction, recently published in Nature Portfolio npj Flexible Electronics in collaboration with the groups of David Maresca and Valeria Gazzola.

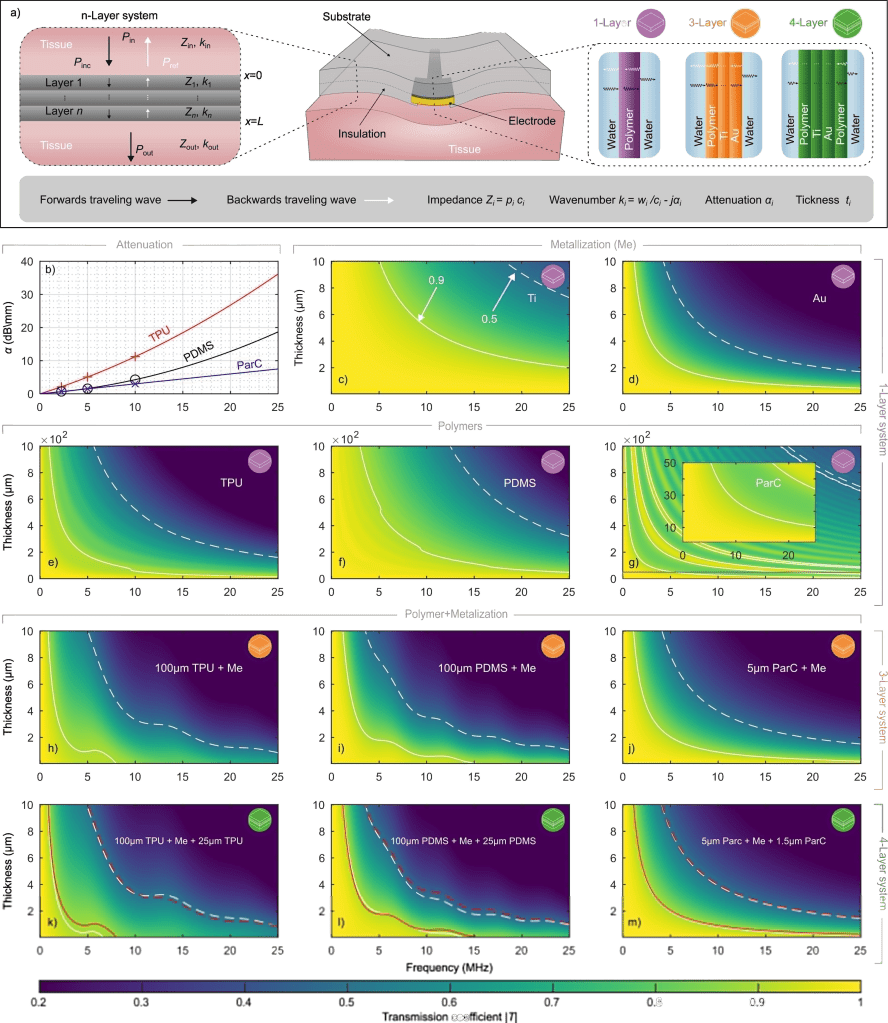
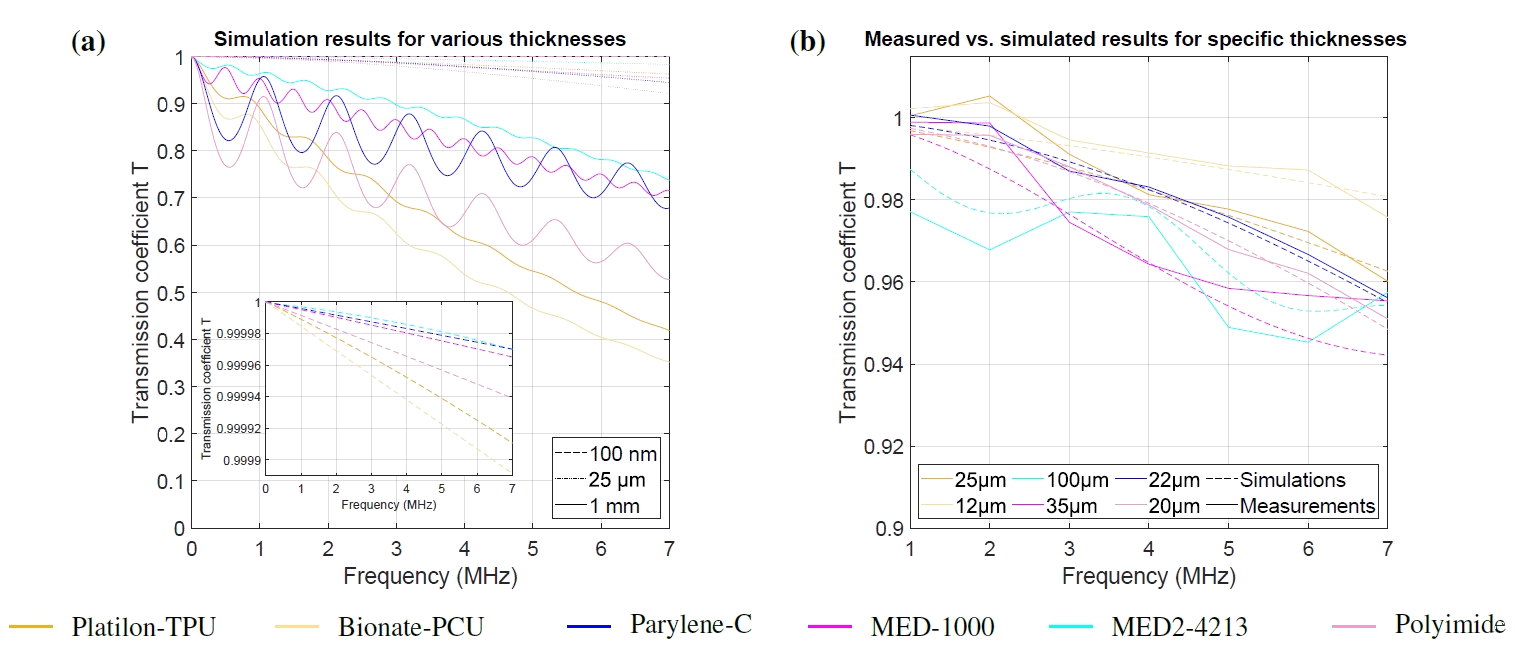
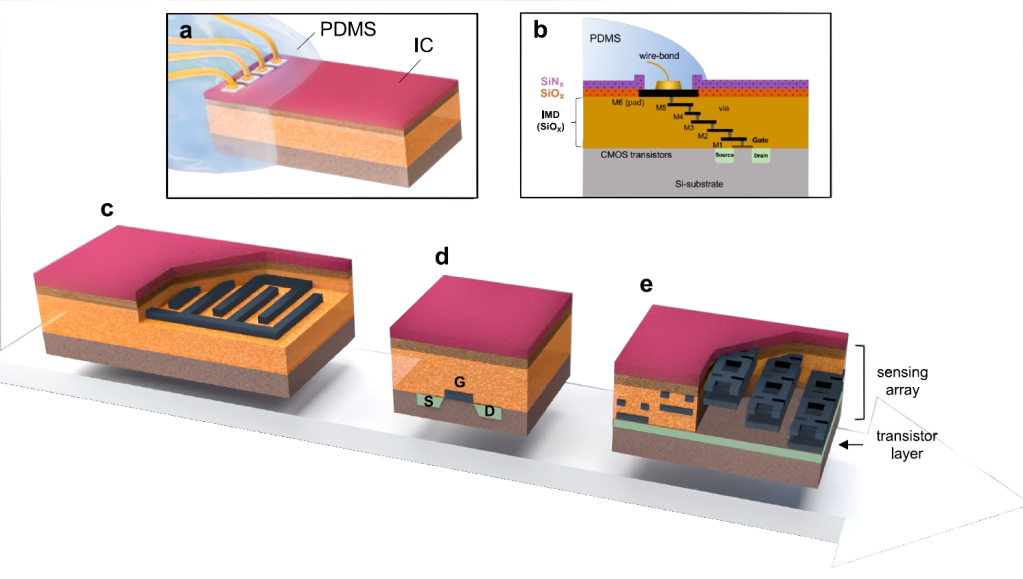
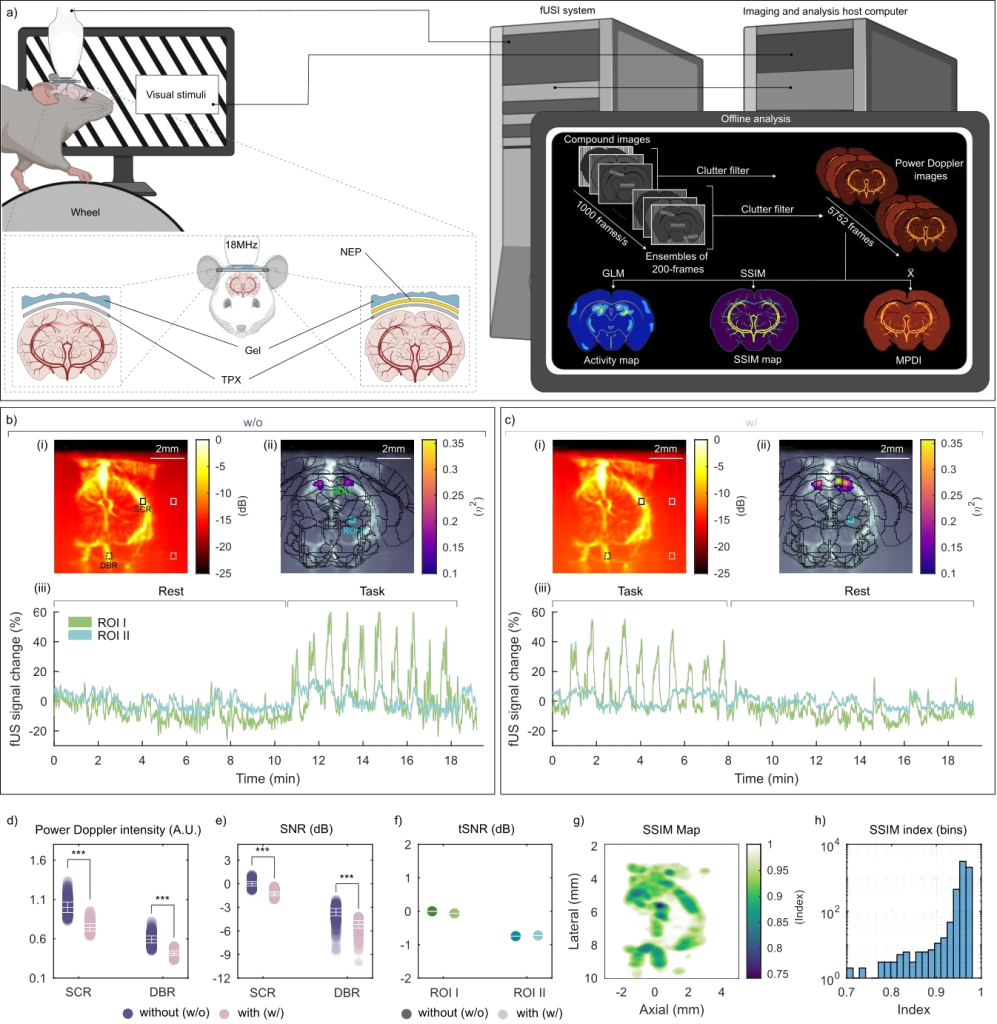
Here, we introduce a framework for designing flexible, metal-based neural interfaces that remain acoustically transparent, enabling integration with functional and focused ultrasound technologies. We combine theory, simulations, and experiments to show how common implant materials and practical metal thicknesses can achieve high ultrasound transmission. Importantly, we demonstrate in vivo compatibility with functional ultrasound imaging (fUSI),
with undistorted signal transmission and robust visualization of subcortical activation through a neural interface. This work lays the foundation for multimodal neural interfaces that bridge electrophysiology, imaging and neuromodulation.
A special shout-out goes to Raphael Panskus, who led this impressive work.
This was an invited contribution to the special collection “Conformable
Brain-Computer and Brain-Machine Interfaces” edited by Xinxia Cai, Jeffrey R Capadona,
Maria Asplund and Ulrich Hofmann.
Cite this paper: R. Panskus, A. I. Velea, L. Holzapfel, C. Pavlou, Q. Li, C. Qin, F. M. Nelissen, R. Waasdorp, D. Maresca, V. Gazzola, and V. Giagka, “Ultrasound Transparent Neural Interfaces for Multimodal Interaction,” npj Flexible Electronics, 2026. doi: 10.1038/s41528-025-00517-1.